September 2018
Distemper Diagnostic Dilemma
By Drs. Kelli Almes and Sara Gonzalez
 |
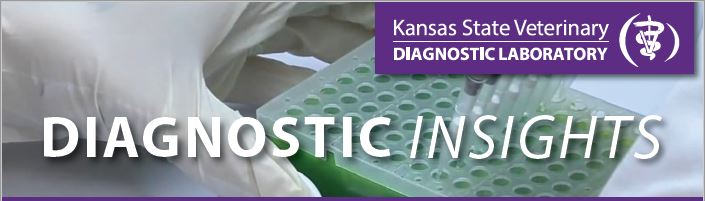
| Figure 1. Green-yellow ocular discharge in a dog with canine distemper virus. |
Diagnosing canine distemper virus (CDV) in a veterinary practice is a high-stakes situation, with high mortality rates and risk of outbreaks. While we hope to prevent distemper through vaccination, we must be prepared to address certain diagnostic dilemmas if we are faced with a potential case.
In order to diagnose distemper antemortem, submitting samples for PCR testing is the best first step. With different clinical manifestations, the appropriate sample type will depend on the primary body system affected.
Vomiting and diarrhea are commonly witnessed with infection. If this is the case for your patient, 2mL of EDTA (purple top) whole blood or feces are preferred specimens for PCR testing. If a dog presents with ocoulonasal discharge (Figure 1), upper respiratory tract infection, or pneumonia, we recommend submitting nasal, pharyngeal, or ocular swabs. Copan e-swabs or sterile swabs placed in a red top tube with 0.25mL of sterile saline are acceptable (gel bacterial swabs are NOT suitable for PCR testing).
 |
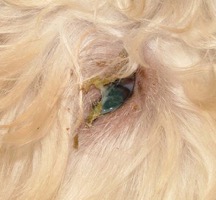
| Figure 2. Diffuse interstitial pneumonia caused by canine distemper virus in a 4-month-old puppy. |
Please watch this video for techniques on collecting a nasal swab. When viremia progresses, neuroinvasion may occur. If your patient exhibits neurologic signs, 2mL of urine (preferred), 2mL EDTA whole blood, or a conjunctival swab provide the best opportunity to obtain a definitive diagnosis.
What if your patient has been vaccinated? Can this interfere with PCR test results? For several weeks after vaccination, CDV PCR can yield positive results. Luckily, we can use the Ct (Cycle threshold) value to interpret the significance of this positive result. Ct values included on your report typically range from the teens to the 30’s. Lower values (teens and low 20’s) are most likely associated with fulminant infection, while higher values (over 30) are an indication of a very early infection, chronic infection, or vaccination.
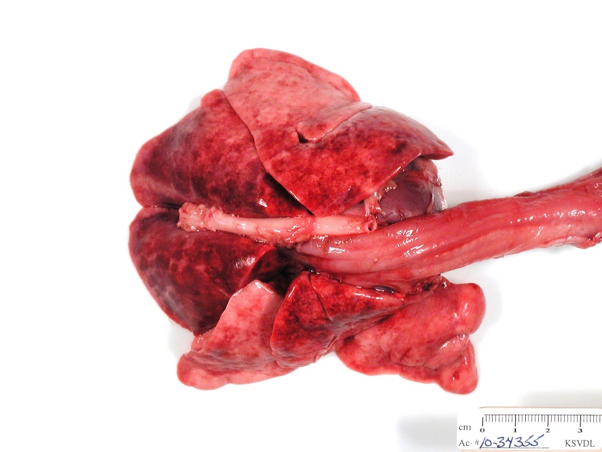
There are many gross and histopathologic lesions that may be found with canine distemper virus, and these lesions are dependent upon the body system affected. This can lead to nonspecific lesions that are often complicated by secondary bacterial infections, especially within the respiratory tract. Pneumonia will often begin as a caudodorsal or diffuse interstitial pneumonia (Figure 2) which progresses to overlapping cranioventral lung consolidation due to secondary bacterial infections. Histologic examination of tissues often reveals characteristic inclusion bodies (Figure 3). If no inclusion bodies are observed, immunohistochemical staining may also be utilized to identify viral antigen within the fixed tissues (Figure 4). Other possible histologic lesions include lymphoid depletion and encephalomyelitis with variable intracytoplasmic and intranuclear inclusion bodies.
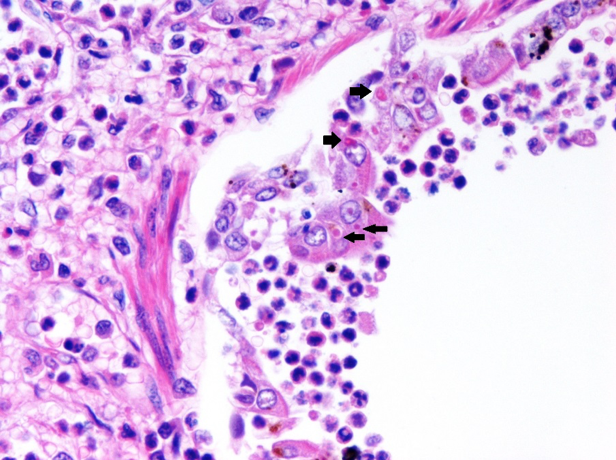 |
| Figure 3. Histopathology of lung with numerous intracytoplasmic viral inclusion bodies (black arrows) in a puppy with canine distemper virus. |
Postmortem diagnostics should include the submission of a full set of fixed and fresh tissues, including brain, submitted to the diagnostic laboratory for confirmatory testing (if the entire animal cannot be submitted for necropsy). Inclusion bodies are most commonly observed in gastrointestinal and urinary epithelium. Chronic cases are most commonly manifested as neurologic disease and histopathology of the brain with IHC staining or brain tissue PCR is recommended. Skin lesions also typically contain inclusion bodies and abundant antigen with IHC staining.
Therapy recommendations are generally supportive. Luckily, CDV is highly susceptible to common disinfectants. Affected dogs should be quarantined 2 weeks past recovery from acute systemic illness, as they may still be shedding virus. Vaccination with modified live products provide strong protection against this disease.
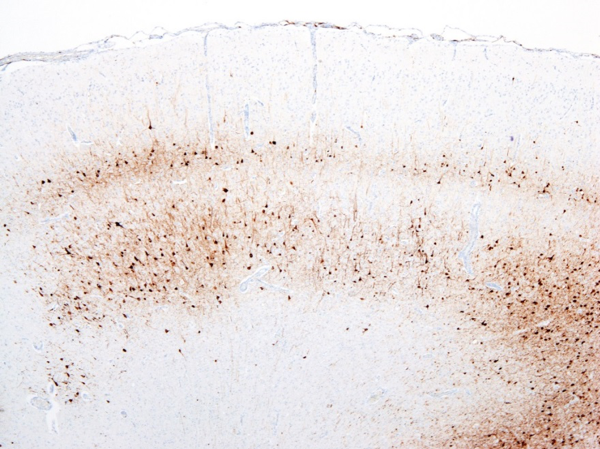 |
|
Figure 4. Immunohistochemical staining for canine distemper virus in the brain of a puppy with neurologic disease. |
NEXT: Canine Cutaneous Plasmacytoma: New Insights
Return to Index