June 2018
Fecal Centrifugation
By Drs. Brian Herrin and Mike Moore
To begin: Mix 2-5 grams of fresh feces (a Hershey’s Kiss weighs 4.5 grams) with approximately 10 ml of floatation solution in a beaker or paper cup.
-
Sugar solution with a 1.27 specific gravity is the best for routine/annual fecal diagnostics
-
Zinc sulfate with a 1.18 specific gravity may be used but it does not float whip worm eggs as well, however it is the solution of choice when you suspect Giardia sp. infections
-
If using a swinging head centrifuge, fill the tube with more solution until you create a positive meniscus above the rim of the tube. Now place the coverslip onto the top and make sure it seals well with the tube. This is the preferred method.
-
If using a fixed head centrifuge, only fill up to approximately 1cm below the rim of the tube.
Place the tube in your centrifuge, balance it, and spin for 5 minutes @ ~ 1300 rpm.
Remove the tube, place it in a rack, and let the tube sit for 10 minutes.
-
This is when more solution should be added if doing the fixed-head method. If you use a squirt bottle for this step, be careful to not cause turbulence in the centrifuge fluid, as this will drive the eggs that are floating deeper into the tube. Add the coverslip and then continue with the 10-minute waiting period.
Remove the cover slip and examine for parasite eggs or oocysts.
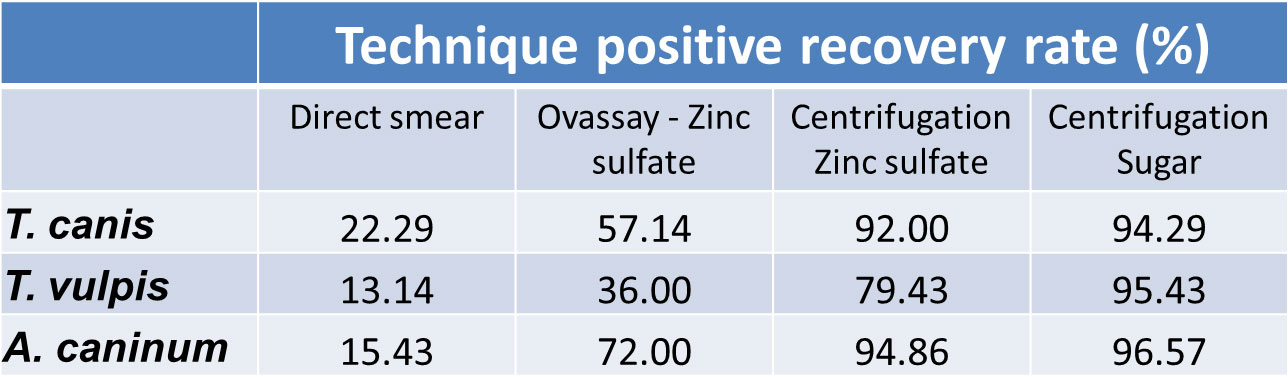
This table demonstrates the improved sensitivity of this technique in the hands of 175 of your colleagues.
To view a short video on this technique go to our You Tube channel @ https://www.youtube.com/watch?v=mZunPcRr7C4&t=0s&list=PLNjV05pK4JEUCABbmIQhKyJzuwk3HbFwF&index=5
If you have questions, please contact KSVDL Client Care at 866-512-5650 or clientcare@vet.k-state.edu