
October 2019
Diagnosing Intestinal Parasites of Dogs and Cats, Part 2
By Dr. Brian Herrin
Like all skills, becoming more comfortable diagnosing parasites in cats and dogs takes practice. We’ve previously discussed how to run a high-quality fecal floatation and how to diagnose roundworms and hookworms. In this article, we will discuss how to diagnose coccidia and giardia.
Whipworms- Trichuris vulpis
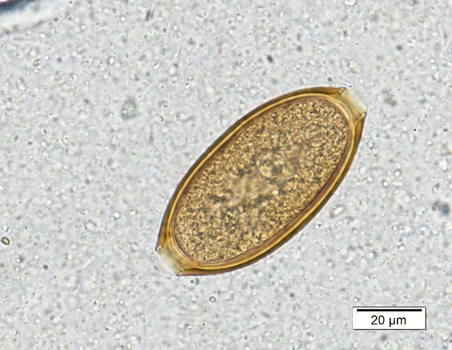
Trichuris vulpis is a common intestinal parasite in adult dogs in Kansas. The eggs of T. vulpis are golden brown, football-shaped with a plug at each end and measure 70 – 90 µm in length by 30 – 40 µm in width (Figure 3a). While the eggs are quite distinct, they do fall into a category of “bipolar-plugged” eggs that can be misidentified.
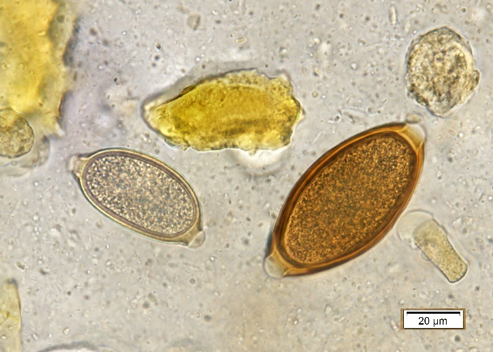
Figure 3b, shows a T. vulpis egg next to a capillarid egg (Eucoleus bohmi). Note that the whipworm eggs are longer than capillarid eggs and are symmetrical along the long axis. This is important as capillarid eggs are either nasal worms or lung worms in dogs instead of true intestinal parasites. In Kansas, a bipolar-plugged egg found in a cat fecal is most likely a capillarid lungworm or a bird capillarid ingested during hunting. There is a Trichuris species that infects cats in southern Florida or the Caribbean, but it is not found outside of that area.
|
|
|
Figure 3: a) Trichuris vulpis egg. Note the golden brown color and bipolar plugs. b) Trichuris vulpis (right) next to a capillarid egg (left). Note that the capillarid egg is smaller, lighter color, and the polar plugs are asymmetric/askew. |
|
Coccidia- Cystoisospora spp.
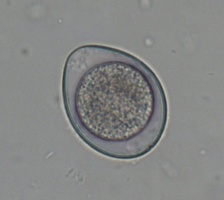
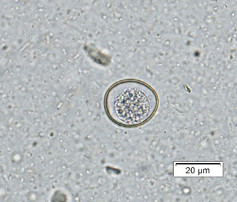
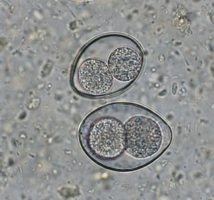
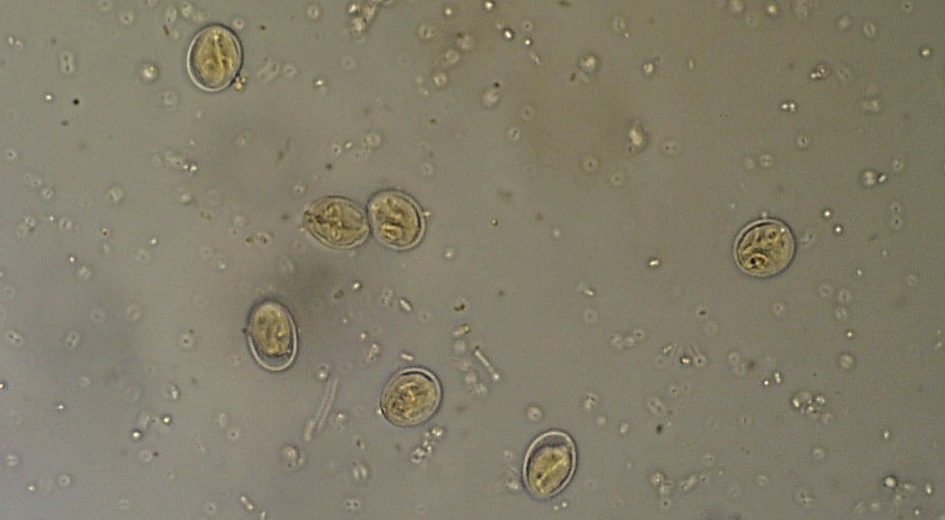
Cystoisospora infections are common in puppies and kittens, especially those in shelter or rescue facilities with a high number of animals. Cystoisospora sp. oocysts have a smooth, clear cyst wall, are elliptical, and contain a single, round cell (unsporulated) (Figure 4a). Both dogs and cats have a “large” coccidium that is approximately 40 – 50 µm in length by 25 – 35 µm in width, and a group of “small” coccidia that are 15 – 25 µm x 15 – 25 µm (Figure 4b). Don’t let this variation in size throw you off; all of these are true parasites of cats and dogs.
The challenge in diagnosing coccidia is that almost all animals have them, and therefore when an animal (mainly dogs) ingests another animal’s feces, those oocysts can pass through, confusing the diagnosis. When differentiating Cystoisospora sp. from Eimeria sp. of ruminants or birds, there are several morphologic characteristics that can help. Some Eimeria spp. have a micropyle cap, which is a small plug at one end of the oocyst. It is not present on all Eimeria sp., so the absence of a micropyle cap is not helpful. Additionally, you can let the sample sit out overnight to further develop, or sporulate. Cystoisospora sp. will sporulate into two sporocysts (Figure 4c) while Eimeria sp. will develop into 4 sporocysts (Figure 4d). The patient can also be held away from the suspected source of fecal ingestion, and the float will be negative within two days. One of the most common sources of pseudoparasites in dogs is Canada goose feces around the neighborhood lakes/parks.
Did you know that IDEXX has found that the most common “parasite” found in the fecal float of adult, healthy dogs is Eimeria spp.? Not a true infection, just passing through after fecal ingestion.
|
|
|
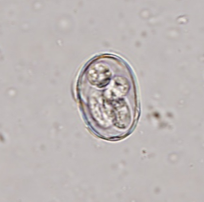 |
| Figure 4: a) Large Cystoisospora sp. oocyst. b) Small Cystoisospora sp. c) Sporulated Cystoisospora sp. Note the two, circular sporocysts. d) Sporulated Eimeria sp. Note the four, overlapping sporocysts within the oocyst. | |||
Giardia sp.
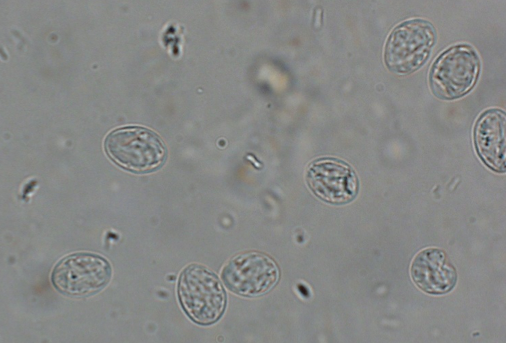
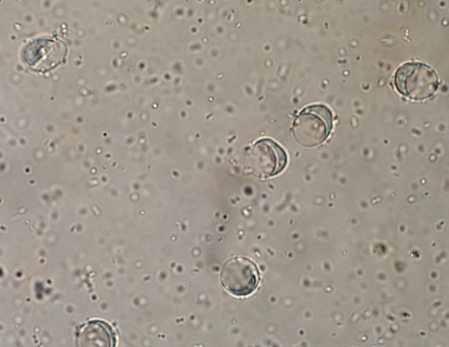
Giardia is one of the more difficult parasites to correctly identify due to its small size and clear color (Figure 5a), but with practice, many veterinary nurses can accurately find cysts on a fecal flotation. The first thing to remember is that a float solution with a lower specific gravity (s.g.= 1.18) must be used. Figure 5b shows what the cysts look like in the wrong float solution. The slide can be stained with iodine to highlight some of the internal structures (Figure 5c).
Giardia cysts are 10 – 15 µm by 7 – 9 µm, and are clear with 2- 4 circular nuclei and a rod running the length of the cyst. When stained, these features are more pronounced. Motile Giardia trophozoites can be seen on direct smears or stained fecal smears as well.
Without proper training, Giardia can be very difficult for technical staff to accurately identify, and therefore the Giardia SNAP test can serve as an alternative diagnostic option.
|
|
|
| Figure 5: a) Unstained Giardia cysts in a zinc sulfate flotation (s.g. 1.18). b) Unstained Giardia cysts in a Sheather’s sugar flotation (s.g. 1.275). c) Giardia sp. cysts stained with Lugol’s iodine. Note the internal structures such as the nuclei and axostyle. | ||
Summary
The goal is to empower the technical staff to recognize the most common parasites that are seen on a daily basis. This takes practice and preparation but can be done in-clinic. Then when unusual/abnormal parasite eggs are seen, technical staff can recognize it is NOT normal and ask for assistance. For those cases of the weird and the wonderful, consulting with book resources or our parasitology team at KSVDL can help with identification. The diagnostic parasitology lab is always willing to help with those challenging cases, including re-running samples, looking at images, or having phone/email consultations. Let us know how we can best serve your clinic’s need!
Did you miss part 1 of Dr. Herrin's article? Find it here.
Next: Non-protein nitrogen ruminant toxicosis
Return to Index