May 2021
Blue-green Algae Poisoning Update
By Dr. Scott Fritz
History:
Blue-green algae (cyanobacteria) are common components of water ecosystems. Uncontrolled “blooms” of these organisms can result in lethal concentrations of cyanotoxins in waters used for recreation, drinking water, and agriculture. (Figure 1 & 2) Lethal blooms are responsible for severe illness and fatalities in nearly all animal species and humans worldwide. In September 2020, blue-green algae were implicated in the fatalities of over 300 elephants in Botswana. Poisonings generally occur during the summer in the U.S. when water temperatures are elevated and the blooms are more severe and of longer duration. While blooms occur in fresh, brackish, and salt waters (usually called “tides” with a color, ie. Red Tide), this article will focus on freshwater blooms in the Midwest.
 |
 |
| Figure 1 | Figure 2 |
| Photos by Dr. Scott Fritz | |
Risk factors:
Risk factors contributing to a severe algal bloom include heavy nutrient concentration (eutrophication), stagnant water, warm water temperatures (<70°F), and warm sunny weather. Nutrient enrichment is commonly associated with residential or agricultural runoff that concentrates nitrogen and phosphorus in a body of water. Many of the toxic algae species are buoyant, allowing them to maximize their photosynthetic abilities. The blooms are often influenced by wind direction and will concentrate on the leeward shore concentrating toxins to a small area of the water body. Animals that drink from this area are the most at risk, and this is the best place to obtain diagnostic samples.
Strains and toxins:
Most cases of toxicity are associated with toxic algal strains within the species Aphanizomenon, Cylindrospermopsis, Dolichospermum (formerly Anabaena), Microcystis, Nodularia, Oscillatoria, and Planktothrix. (Figure 3) Most cyanobacteria are capable of producing multiple cyanotoxins and blooms are commonly a mixture of different algal species confounding risk assessments. Clinically, cyanotoxins are best separated into hepatotoxic and neurotoxic varieties.
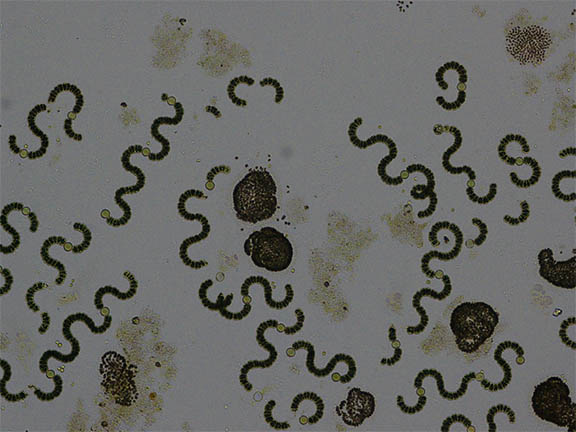 |
| Figure 3 |
The most commonly encountered cyanotoxin in water samples submitted to KSVDL is microcystin and is usually associated with the algae Microcystis. Microcystins are potent hepatotoxins that inhibit protein phosphatases leading to hepatocyte infrastructure reorganization and eventual apoptosis of hepatocytes. Apoptosis of hepatocytes leads to intrahepatic hemorrhage and hypovolemic shock. Clinical signs initially reflect gastrointestinal insult, vomiting, bloody diarrhea, and colic. Signs will progress to lethargy, weakness, pallor, seizures, and death (hours to days) as the liver is damaged.
Surviving animals may have impaired liver function. Cattle and horses that survive the initial insult may suffer from photosensitization. KSVDL also identifies Dolichospermum regularly in water samples. This species is capable of producing microcystin as well as the neurotoxin: anatoxin-a. Anatoxin-a is a potent post-synaptic cholinergic agonist that causes a depolarizing neuromuscular blockade. Clinical signs associated with anatoxin-a exposure include tremors, seizures, convulsions, and death due to respiratory failure (minutes to hours). Anatoxin-a(s) is another neurotoxin that inhibits acetylcholinesterase causing SLUDD syndrome. Lesions associated with the neurotoxins are non-specific and often not present at all due to the rapid progression to death. Both classes of algal toxins have steep dose-response curves, research has shown ingestion of 90% of a lethal dose does not result in outright signs of poisoning.
Diagnosis:
Diagnosis is a combination of history of exposure, clinical signs, and necropsy findings. There are analytical techniques to measure these toxins in water samples KSVDL can identify algae in water samples. via light microscopy (Blue-Green Algae: TOX-1003) and can also measure the amount of toxins present in suspect water (Microscystin/Cylindrospermopsin: TOX-2101). . Please follow this link for guidance on water sampling procedures: (Correct water sampling guidance)
Treatment:
Treatment can be frustrating. Many times owners do not realize there was an exposure and affected animals are not examined until they exhibit clinical signs. By this time decontamination efforts are of little value. Whereas emesis followed with activated charcoal and a cathartic immediately after consumption will likely be curative. Supportive care of clinical patients is imperative. Current therapy recommendations for cases of hepatotoxicosis in companion animals include: cholestyramine, IV fluids, antiemetics, and potentially antibiotics to prevent secondary infections. Whole blood transfusions may be indicated in severe cases. Therapy recommendations for cases of anatoxin-a are limited in the literature. These cases are often so rapidly fatal that veterinary intervention isn’t possible. Atropine may reduce some of the clinical signs associated with anatoxin-a(s) exposure.
The best treatment is preventing exposure. Client education is an important aspect of prevention. Animal owners may be unaware of the potency of these toxins and may not realize the very real risk their animals face in a water body with an active bloom. Many state agencies monitor recreational waters and post warning signs at access points. The advisories for Kansas can be found at: Kansas Department of Health and Environment Aglal Bloom Advisories.
When warning signs are posted at large water bodies in your area, the conditions are likely ideal for blooms in smaller private ponds. Owners that notice a paint-like scum on the surface of water or a rapid color change should avoid using that water for animal drinking water or recreation. (Figures 1 & 2) Water samples can be submitted to KSVDL for testing, the ideal sample is <100mL of water taken from the down-wind side of a pond ~one inch below the surface. The sample can be shipped on ice, do not freeze the sample as this can lyse algal cells complicating identification. (Correct water sampling guidance)